WENDELL ROELF
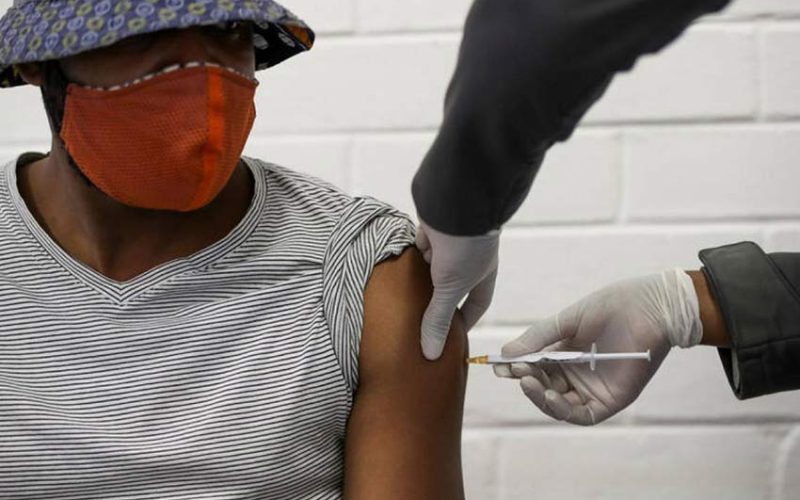
SOUTH Africa aims to immunise between 350,000 and 500,000 health workers with Johnson & Johnson’s COVID-19 vaccine in an “implementation study” to further evaluate the shot, the president of the country’s Medical Research Council said.
Glenda Gray, co-lead investigator on the local leg of a J&J global trial, told Reuters South Africa expected to get batches of around 80,000 doses every seven to 14 days for the study, once it is approved.
The implementation study would be aimed at further evaluating J&J’s vaccine in the field and would be akin to a phase IIIb study, Gray said. J&J’s vaccine has already been tested in the global phase III trial involving more than 40,000 participants including over 6,000 in South Africa.
Gray’s comments come days after the government paused the rollout of 1 million doses of AstraZeneca’s vaccine and switched to the J&J shot to start protecting its health workers.
That decision was based on a small clinical trial that showed the AstraZeneca jab provided minimal protection against mild to moderate illness from the more contagious 501Y.V2 virus variant that is now dominant in the country. The government said on Wednesday it was considering selling or swapping AstraZeneca shots it had ordered.
Health Minister Zweli Mkhize said earlier on Wednesday that the J&J implementation study would “provide valuable information about the pandemic in the post-vaccination community and thus, ensure early identification of breakthrough infections should they occur amongst vaccinated health workers”.
Gray said: “We will get 80,000 (doses) every 7 to 14 days and then based on our ability to roll them out we will redeploy them.”
She added that a critical next step was for J&J to get emergency use authorisation (EUA) in the United States for its vaccine. The U.S. Food and Drug Administration (FDA) is expected to meet on Feb. 26 to discuss J&J’s EUA request.
“Once the FDA has approved it, there’s a cascading process that goes to other countries,” Gray said.
“South Africa has a bilateral agreement with the U.S. FDA and they could accept the EUA from another agency. Alternatively, South Africa could wait until there is a formal process directly with J&J and (medicines regulator) SAHPRA.”